Researchers from North Carolina State University, the University of North Carolina at Chapel Hill and Laser Zentrum Hannover have discovered that a naturally-occurring compound can be incorporated into three-dimensional (3-D) printing processes to create medical implants out of non-toxic polymers. The compound is riboflavin, which is better known as vitamin B2.
“This opens the door to a much wider range of biocompatible implant materials, which can be used to develop customized implant designs using 3-D printing technology,” says Dr. Roger Narayan, senior author of a paper describing the work and a professor in the joint biomedical engineering department at NC State and UNC-Chapel Hill.
The researchers in this study focused on a 3-D printing technique called two-photon polymerization, because this technique can be used to create small objects with detailed features – such as scaffolds for tissue engineering, microneedles or other implantable drug-delivery devices.
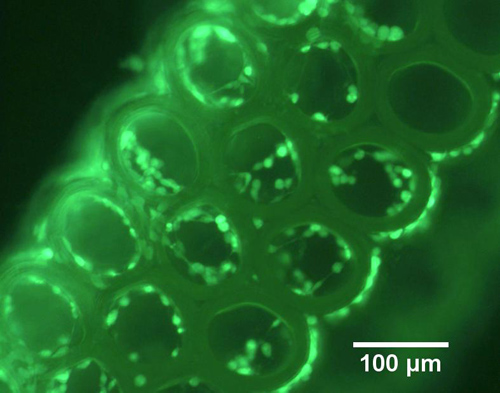
The discovery enables researchers to create finely-detailed, biocompatible structures, such as this scaffold for tissue engineering. Image credit: Image Citation: Regen. Med. 8(6), 725–738 (2013).
Two-photon polymerization is a 3-D printing technique for making small-scale solid structures from many types of photoreactive liquid precursors. The liquid precursors contain chemicals that react to light, turning the liquid into a solid polymer. By exposing the liquid precursor to targeted amounts of light, the technique allows users to “print” 3-D objects.
Two-photon polymerization has its drawbacks, however. Most chemicals mixed into the precursors to make them photoreactive are also toxic, which could be problematic if the structures are used in a medical implant or are in direct contact with the body.
But now researchers have determined that riboflavin can be mixed with a precursor material to make it photoreactive. And riboflavin is both nontoxic and biocompatible – it’s a vitamin found in everything from asparagus to cottage cheese.
The paper, “Two-photon polymerization of polyethylene glycol diacrylate scaffolds with riboflavin and triethanolamine used as a water-soluble photoiniator,” is published online in Regenerative Medicine. Lead author of the paper is Alexander Nguyen, a Ph.D. student in NC State and UNC-Chapel Hill’s joint biomedical engineering program. Co-authors include Shaun Gittard, Anastasia Koroleva, Sabrina Schlie, Arune Gaidukeviciute and Boris Chichkov of Laser Zentrum Hannover. The research was supported by National Science Foundation grant 0936110.
*Source: North Carolina State University
