Washington, D.C.— Glasses differ from crystals. Crystals are organized in repeating patterns that extend in every direction. Glasses lack this strict organization, but do sometimes demonstrate order among neighboring atoms. New research from Carnegie’s Geophysical Laboratory reveals the possibility of creating a metallic glass that is organized on a larger scale. Their results are published June 17 in Science.
Scientists have discovered glasses that demonstrate order among the nearest neighboring atoms, called short-range order, and a slightly wider range of atoms, called medium-range order. Most research about finding or creating a glass with a long-range, nearly crystalline, level of order—referred to as the perfect glass state—has been conducted on ice and the minerals silica and zeolite. But no research into long-range order glass has been successful until now.
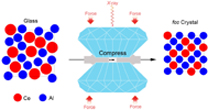
An illustration of how the atoms in cerium-aluminum glass become ordered and develop a crystalline structure under pressure. Image credit: Carnegie Institution for Science
The research team, including Carnegie’s Ho-Kwang (Dave) Mao, focused on metallic glass made from the elements cerium and aluminum. Metallic glasses are a hot research topic, because they are less brittle than ordinary glasses and more resilient than conventional metals. They combine the advantages and avoid many of the problems of normal metals and glasses, two classes of materials with a very wide range of potential applications.
By placing the cerium-aluminum glass under 25 gigapascals of pressure–about 250,000 times normal atmospheric pressure–the team was able to create a single crystal. When the glass was brought back to ambient pressure, the new structural order was preserved.
Using x-ray techniques and simulations, the team determined that the atomic structures of cerium and aluminum prevent the glass from assuming the highly ordered state at normal pressures. But under the intense 25 gigapascals, an electron in cerium shifts, allowing the crystalline structure to be created.
“These exciting results demonstrate that pressurized cerium-aluminum glass could be a favorablesystem for discovering the long-sought-after perfect glass,” Mao said. “This situation could also exist in other metallic glasses.”
*Source: Carnegie Institution for Science
